Coronavirus: Oxford Uni reveals details of COVID-19 vaccine trial – here's how to get involved
Testing for a potential coronavirus vaccine begins on Thursday and details have been revealed on how to volunteer to take part.
Health secretary Matt Hancock announced on Tuesday that Oxford University will begin trials on humans this week, pledging more than £40m in funding to help the process.
Now Oxford University has revealed how and where people can can put themselves forward to be involved – and that they will be paid.

The university is asking for healthy adults aged between 18 and 55 who have not tested positive for COVID-19 for the trials, which will last for a total of six months.
Those selected will be reimbursed for their time, which the university says will mean anything from £190 to £625.
Latest coronavirus news, updates and advice
Live: Follow all the latest updates from the UK and around the world
Fact-checker: The number of COVID-19 cases in your local area
6 charts and maps that explain how COVID-19 is spreading
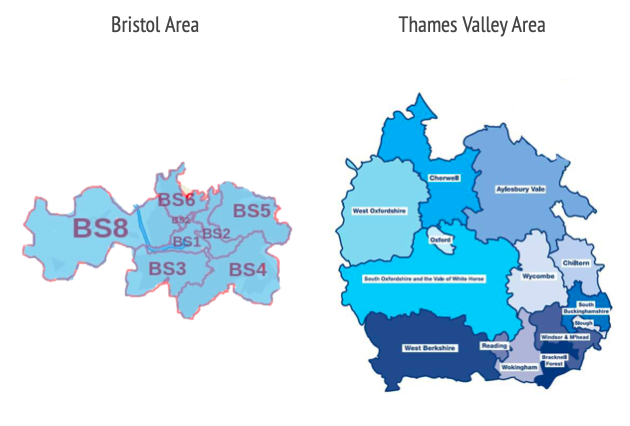
The selection process is not available for the whole country. Only people living in specific postcodes in Bristol, Thames Valley, Southampton and Greater London will be eligible.
Announcing the government’s support for the trials, Hancock said: ”In the long run, the best way to defeat coronavirus is through a vaccine.
"After all, this is a new disease, this is uncertain science, but I'm certain we will throw everything we've got at developing a vaccine.
The health secretary acknowledged the process for finding a vaccine would take "trial and error", but told UK scientists leading the search he would "back them to the hilt and give them every resource they need" in order to succeed.

Work on the vaccine, developed by clinical teams at Oxford University's Jenner Institute and Oxford Vaccine Group, began in January.
The UK will join the US – which has two studies – Germany and China, in beginning human trials.
Elsewhere, in a separate study at Imperial College London, researchers have developed a vaccine candidate that, when injected, will deliver the genetic instructions to muscle cells to make the SARS-CoV-2 spike surface protein.
This should provoke an immune response and create immunity to the virus.
The team, led by Professor Robin Shattock from Imperial's Department of Infectious Disease, has been testing the candidate in animals since early February.
Clinical trials are expected to begin in June and the team will look to recruit healthy adults to test the vaccine.
Results could be available as soon as September, the researchers say.
Coronavirus: what happened today
Click here to sign up to the latest news, advice and information with our daily Catch-up newsletter



